You should know that the matter consists of very small structural units known as the molecules .
The molecules
You should know that the molecule is the smallest part of the matter which can exist freely , and it has the properties of the matter .
The properties of the molecules of the matter
You should know that the molecules of the matter are in a continuous motion , and there are intermolecular spaces and attraction forces among the molecules of the matter .
The intermolecular spaces are the spaces that are found among the molecules of the matter .
The intermolecular force is the force that bounds the molecules of the matter together .
You should know that the molecules of one substance are alike in properties , but they differ from the other substance molecules . The molecules are composed of tiny structural units called the atoms .
You should know that the difference in the molecules of the various substances is found as the result of the difference of the atoms which are involved in the structure of the molecule , and depends on the number and the kind of the atoms , and the way of the combination together .
The kinds of the molecules are the element , the compound and the mixture .
The element molecule
You should know that the element is the simplest pure form of the matter which can not analyzed chemically into the simpler form . The element molecule is formed of the similar atoms ( one or more atoms ) that combine together .
 |
| The monoatomic elements are elements formed of one atom , the diatomic elements are elements formed of two atoms . |
Monoatomic elements
There are elements molecules that formed of only one atom ( The Monoatomic ) as
The solids as ( Copper , Iron , Aluminum , Sulphur , Magnesium and Carbon ) .
The liquids as mercury .
The nobel gases such as ( Helium , Neon , Argon , Krypton , Xenon and Radon ) .

The nobel gases are monoatomic .

Diatomic elements
There are elements molecules that formed of two atoms ( Diatomic ) . such as The liquids as bromine .
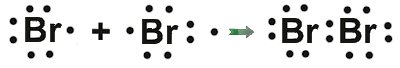 |
| Bromine is the liquid element which is composed of two atoms . |
The active gases as ( Oxygen , Hydrogen , Chlorine , Nitrogen and Fluorine .
 |
| Oxygen molecule is an active gas , and the molecules of the active gases are diatomic . |
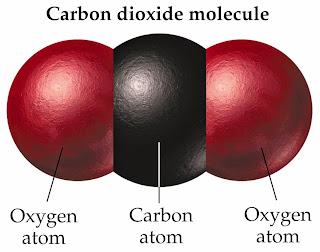
The compound molecule
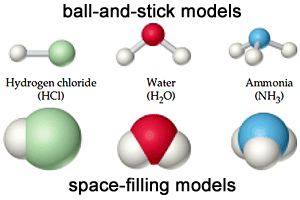 |
| The compound molecule is formed from different atoms . |
No comments:
Post a Comment